Fresh Study Unveils Crucial Stages in the Assembly and Recruitment of Lassa Virus Ribonucleoparticles
Lassa virus (LASV) is the causative agent of Lassa hemorrhagic fever, a disease endemic to West Africa, resulting in approximately 5,000 fatalities annually. Collaboratively, the Uetrecht group from CSSB Centre for Structural Systems Biology (CSSB, LIV, Uni Siegen), the Kosinski group from CSSB and EMBL, and the Rosenthal group from BNITM and CSSB, have elucidated the pivotal role of RNA in essential stages of the Lassa virus life cycle.Within the human body, more than 20,000 genes produce over a million diverse protein variants. In contrast, the Lassa virus is remarkably concise, consisting of only four proteins—L, NP, Z, and GPC.
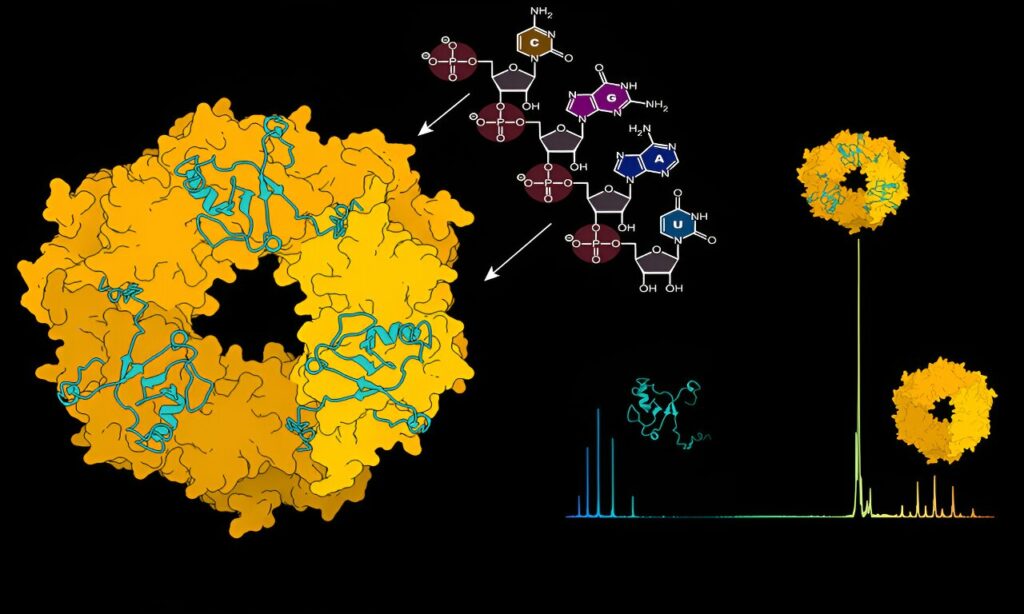
“We aim to comprehend how these four proteins can induce severe damage to human cells,” notes Lennart Sänger, the primary author of the paper. “The activities and expression of these proteins must undergo precise regulation, and efficient communication among them is essential for undertaking various functions.”
To evade detection by the immune system, the nucleoprotein (NP) envelops the viral genome in a capsid. This capsid, in conjunction with viral RNA and the L protein, constitutes ribonucleoprotein complexes (RNPs).For infection propagation, RNPs must continually restructure to facilitate viral genome replication and transcription. The researchers delved into the interactions between NP and viral RNA, along with the Z protein, seeking a deeper understanding of the mechanism and dynamics of RNP formation and incorporation into new viral particles.
Utilizing structural mass spectrometry, a method akin to a molecular scale that unveils the atomic weight of molecular interactions, the researchers scrutinized the dynamics between NP and viral RNA. Charlotte Uetrecht, a CSSB group leader and an expert in mass spectrometry techniques, elucidates, “Initially, the NP protein lacks a composition capable of binding viral RNA. A transformation must occur to enable this binding, and we found that viral RNA can autonomously initiate this transformation.” The researchers identified RNA as the catalyst for the disassembly of ring-like NP trimers into monomers, which can then assemble into higher-order RNA-bound NP structures.
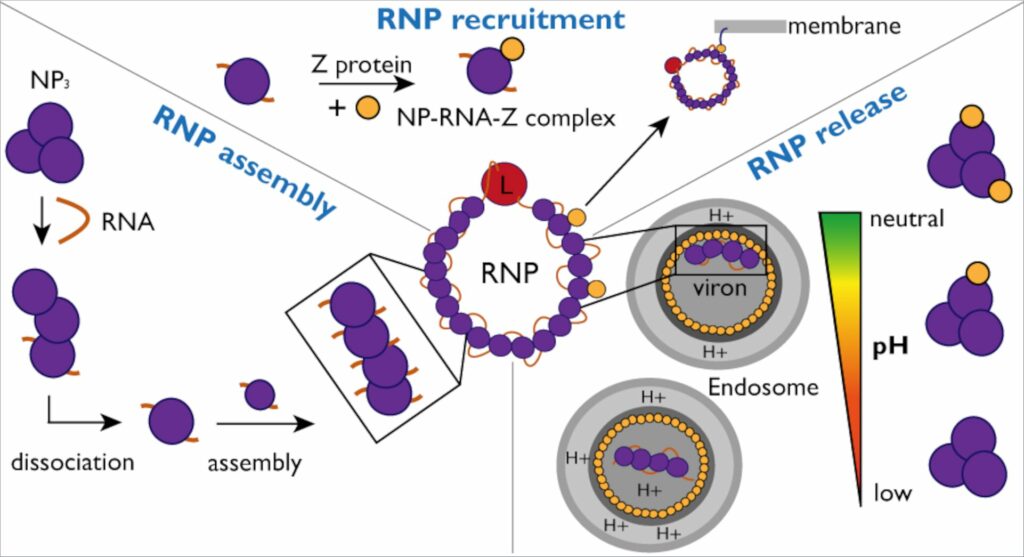
The investigators also delved into the NP interaction with the Z protein in greater detail. To facilitate this examination, the Kosinski group utilized AlphaFold to predict the interaction site of the NP-Z complex. These predictions were subsequently validated through laboratory experiments.”
Employing artificial intelligence allowed us to swiftly identify potential interactions and create mutants to validate our hypotheses,” remarks Jan Kosinski. The researchers successfully demonstrated that while NP binds with Z independently of RNA presence, this interaction is contingent on pH.”In essence, these findings enhance our comprehension of RNP assembly, recruitment, and release in the Lassa virus,” elucidates Maria Rosenthal, an expert on Lassa virus at the Bernhard Nocht Institute for Tropical Medicine and a CSSB associate member. In West Africa, approximately 186 million people are anticipated to be at risk of Lassa virus infection by 2030, and the World Health Organization acknowledges Lassa virus as a perilous and yet insufficiently studied pathogen.”
Understanding the functioning of the Lassa virus may ultimately pave the way for the development of molecules that could impede the virus’s replication and treat Lassa fever,” Rosenthal adds.
This article is republished from PhysORG under a Creative Commons license. Read the original article.
Do not forget to share your opinion with us to provide you with the best posts !



0 Comments