Discovery of Water Molecules Challenges Conventional Textbook Models
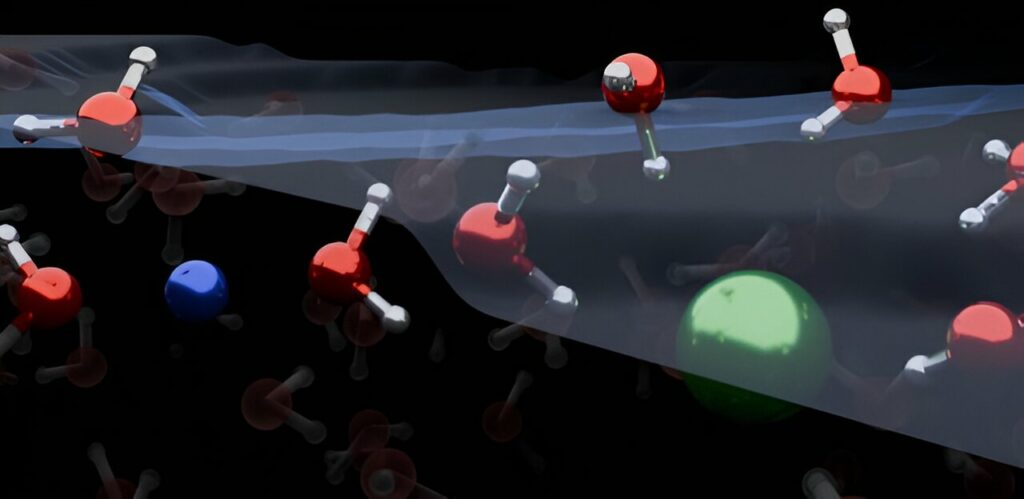
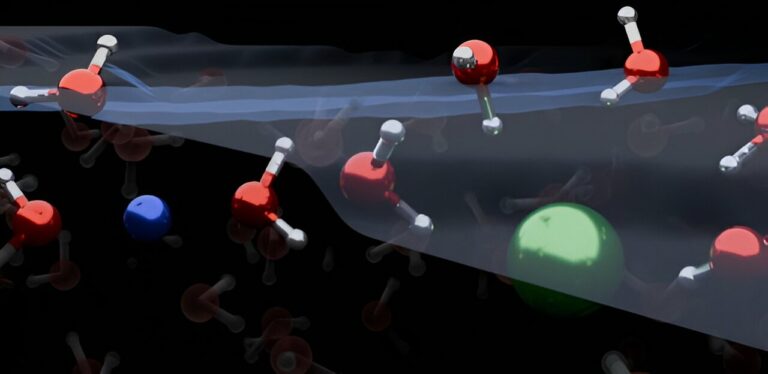
The organization of water molecules at the surface of salt water has been found to be different than previously believed, requiring a re-drawing of textbook models. These surface reactions between water and air are crucial for understanding climate and environmental processes, such as ocean water evaporation and its impact on atmospheric chemistry and climate science.
The distribution of ions at the air-water interface can significantly influence atmospheric processes, but the microscopic reactions at these interfaces have been a subject of intense debate. However, a recent study published in Nature Chemistry by researchers from the University of Cambridge and the Max Planck Institute for Polymer Research in Germany reveals that the organization of ions and water molecules at the surface of electrolyte solutions, like most salt-water solutions, is fundamentally different from what was traditionally believed. This discovery has the potential to improve atmospheric chemistry models and other related applications.
A more sophisticated technique
The objective of the researchers was to investigate the impact of ion distribution on water molecules at the precise boundary between air and water. Traditionally, this has been accomplished using vibrational sum-frequency generation (VSFG), a laser radiation technique that directly measures molecular vibrations at these critical interfaces.
However, while the strength of the signals can be measured, the technique does not determine whether the signals are positive or negative, making it challenging to interpret previous findings. Moreover, relying solely on experimental data can lead to ambiguous results.
To overcome these challenges, the team employed a more advanced form of VSFG known as heterodyne-detected (HD)-VSFG to examine various electrolyte solutions. They then developed sophisticated computer models to simulate different interface scenarios.
The combined findings revealed that both positively charged ions (cations) and negatively charged ions (anions) are depleted from the water/air interface. Simple electrolytes cause water molecules to orient in both upward and downward directions. This contradicts conventional models, which suggest that ions form an electrical double layer and only orient water molecules in one direction.
Dr. Yair Litman, co-first author from the Yusuf Hamied Department of Chemistry, stated, “Our research demonstrates that the ion distribution at the surface of simple electrolyte solutions differs from previous assumptions, and the ion-enriched subsurface determines the organization of the interface: a few layers of pure water at the top, followed by an ion-rich layer, and finally the bulk salt solution.”
Co-first author Dr. Kuo-Yang Chiang of the Max Planck Institute added, “This study highlights the invaluable contribution of combining high-level HD-VSFG with simulations in enhancing our understanding of liquid interfaces at the molecular level.”
Professor Mischa Bonn, who leads the Molecular Spectroscopy department,
This article is republished from PhysORG under a Creative Commons license. Read the original article.
Do not forget to share your opinion with us to provide you with the best posts !



0 Comments