Creating a synthetic derivative of natural indigo could serve as a catalyst for innovation in future electronic devices.
RIKEN chemists have developed a method to create synthetic versions of indigo dye without the need for harsh conditions. This breakthrough could have significant implications for the advancement of electronic devices, such as light-responsive gadgets and flexible biomedical sensors.
The interest in semiconductors based on organic molecules is growing due to their flexibility, lightweight nature, and ability to be molded. Unlike traditional silicon-based semiconductors, organic molecules offer a wide range of structural possibilities. Keisuke Tajima, the lead researcher at RIKEN Center for Emergent Matter Science, explains that organic semiconductors can be designed with new functionalities due to their molecular flexibility.
To explore the potential of molecular design in enhancing electronic function, Tajima and his team focused on a molecule similar to indigo called 3,3-dihydroxy-2,2-biindan-1,1-dione (BIT). Their research aimed to determine if protons and electrons could move together in the solid state. Proton-coupled electron transfer, which links the movement of electrons to protons, is crucial for efficient electron transfer in biological systems. If this phenomenon can be incorporated into organic solid-state materials, it could result in semiconductors with unique dynamic properties. However, until now, no solid-state material capable of displaying proton-coupled electron transfer had been demonstrated.
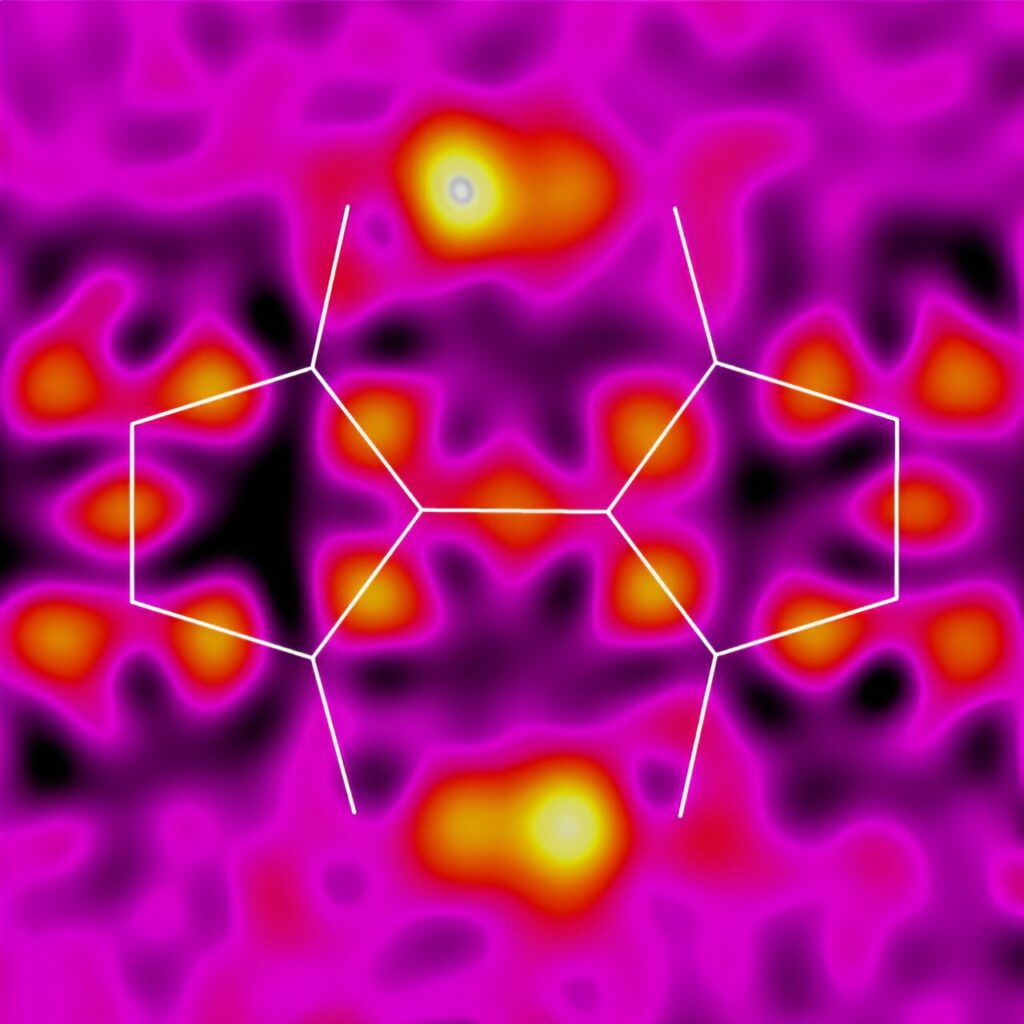
Tajima and his team have made an interesting discovery regarding BIT and its derivatives. They have found that these materials undergo unique rearrangements in their structures, involving double-proton transfer. This discovery suggests that BIT and its derivatives may possess special capabilities as electronic functional materials.
Tajima initially identified BIT and its derivatives as promising materials for solid-state proton-coupled electron transfer. This is because the molecule contains two protons that are strategically positioned to facilitate the transfer of electrons from one location to another.
In the past, the production of BIT required harsh conditions, which limited the range of derivatives that could be synthesized. However, Tajima’s team has developed a new approach that allows for the synthesis of several BIT derivatives under much milder conditions, at room temperature.
With these BIT derivatives in hand, the team delved into studying their properties. One of the challenges they faced was proving that the protons in BIT undergo proton transfer between molecules in the solid state. To overcome this, they collaborated with experts in X-ray crystallography and solid-state nuclear magnetic resonance (NMR) from RIKEN. Through their joint efforts, the team successfully demonstrated that the two protons in BIT do exchange their positions rapidly.
Based on calculations, it is believed that this proton transfer is coupled with charge transport. The team’s next objective is to experimentally confirm this coupling. Tajima explains that while they are uncertain if the presence of a proton will enhance charge transport, it presents an intriguing avenue for further exploration in fundamental physics.
This article is republished from PhysORG under a Creative Commons license. Read the original article.
Do not forget to share your opinion with us to provide you with the best posts !



0 Comments